
Return to Lesson Plan Index
Printer Friendly Version
Grades: 8-10
Author: Jon Valasek
Source: This lesson was inspired by information given me by Debbie Goodwin, material science instructor and Doug Rummel, 8th grade science teacher at St. Mark's School of Texas.
This activity involves the students in an inquiry into classifying various materials.
What should students know as a result of this lesson?
What should the students be able to do as a result of this lesson?
You will need to gather household items that include: plastics (polymer recycling codes 1-7), metals (aluminum, iron, etc.), glass (bottles and jars), compounds (salt, sugar, baking soda, etc.), solutions (colored flavor drinks, tea, coffee, etc), and a variety of materials that are heterogeneous (granite, rocks, wood, etc.). You should supply enough items so that each student has at least one item and distribute at least four or five items per material category. Provide some discrepant items such as a light bulb or capped bottle which are assemblies and do not fit any one category.
Engagement
Before giving materials to students to handle, remind them to exercise safe procedures. Introduce the lesson by passing out the items you have collected; one to each student. Ask, "Each one of these items is a piece of matter because? Solicit responses. (Answer is that matter has mass and occupies space.) Ask, "How would you describe your item?" Accept all responses. (Example: A student holds up an aluminum wire and states that it is solid, shiny, and the same throughout. List each response on the board. Give students an opportunity to look over all the responses. Ask, "Could we take these responses and organize them into categories?" After the students offer suggestions, say, "Look at all the entries on the board and group items that have the same thing in common." An example would be all things that are shiny and solid should be grouped together as "metals".
Assessment: Have students work in groups to organize all the items that have been distributed into categories.
Exploration
Handout or put on the board a list of terms: matter, states, materials, heterogeneous, homogeneous, solids, liquids, gases, solutions, pure substances, element, atoms, and compounds. Using the diagram below take one of the objects and ask a series of questions that leads students to operationally define the terms. "Is this matter? What state is it? Is it uniform (homogeneous)? Is it an element?" You are looking for descriptions that fit the ones in the Content knowledge section (see below). Then using these terms have the students develop a hierarchy of these terms. You might lead the students through the diagram or have them draw a concept map. See AGPA links. Their results should look like:
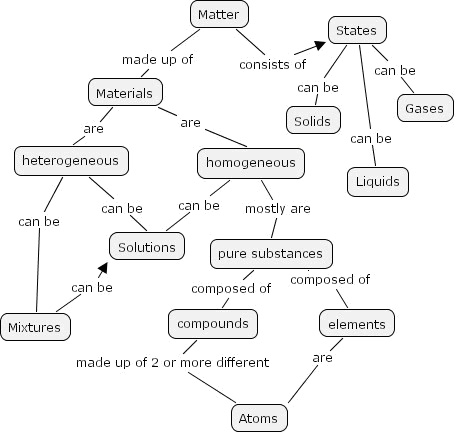
Assessment: Have students share their maps with others in their group. Members of the group should perform self-assessment to agree on one concept map, which they will later share with the whole class.
Explanation
Have each group share their results with the entire class. State that we can categorize all matter by the use of this diagram but there is another scheme that is more practicable. It involves four categories metals, ceramics, polymers, and composites. Without defining each have the students place their object in one of the four categories and state the reasons for their selection. Note the student's selection on the board.
Assessment: After each object is placed ask if any should be moved. In summary ask for the definition for each category.
Elaboration
As a final exercise have the students take their object and integrate it into the diagram previously created. Note that the students must first integrate the four material categories into the diagram first, and then place their object onto the diagram. See a possible solution below:
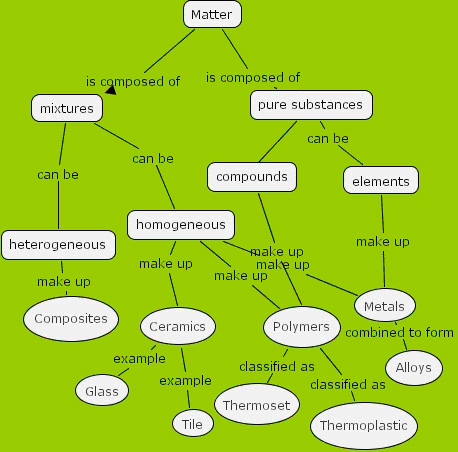
Assessment: Check student's diagram for accuracy.
Knowledge of concept mapping would help but the exercise can be conducted without this knowledge.
NGSS Standards:
Common Core Standards:
National Standards:
Ohio Standards:
Heterogeneous- is not uniform when examined by sight. Tree bark would be heterogeneous.
Homogeneous- is uniform when examined by sight. Unspoiled milk is homogeneous.
Materials can be heterogeneous or heterogeneous and are made up of the following categories:
Elements- A substance that cannot be broken down by physical or chemical means.
Compounds- A combination of two or more elements that can be broken down to elements.

Choose items that can be safely handled in the classroom. Remind students to handle all materials safely and for their intended purpose.
Knowledge of matter and materials promotes scientific literacy among students. Also, these materials show up in thousands of places/things - engineers and scientists choose materials for use based on the materials' physical properties. Classification is an important higher order skill that allows an individual make some sense out of our complex world.
Students should be able to systematically use their observations to classify any substance.
Grouping Suggestions: Students should be placed in groups of two or three. However a novel idea on grouping is found in, "Cooperative Learning in the Science Classroom," by Emily Lin, the Science Teacher Vol 73 No 5 Summer 2006, pp34-39
Pacing/Suggested Time: Two 45 minute periods.
N/A - Students devise their own diagrams.